Import Alert Imposed During Natural Products Association CEO’s Tenure at FDA

Update: As of 2022 NPA and Daniel Fabricant appear to be no longer to be in favor of kratom prohibition***
The Natural Products Association (NPA) describes itself as the “leading trade association for dietary supplements”. Its board members include representatives from massive “nutraceuticals” manufacturers, big names like GNC, Vitamin Shoppe, and Nature’s Way (subsidiary of pharma giants Schwabe), Danisco (subsidiary of Dupont), and chemical companies like Mitsubishi Gas Chemical America and BASF, among others. NPA’s ‘About‘ page informs us that “In 1994, NPA played a key role in the passage of the Dietary Supplement Health and Education Act (DSHEA).”
With a $117 billion industry, you’d think those in the “natural products” world would love to profit from a plant that in its most natural form actually works. For the past decade, the NPA has instead been active and outspoken in its opposition to kratom. The Big Dietary Supplements industry, like its twin industry in Big Pharma (in many cases they’re one in the same), has been trying to stop kratom entirely.
The FDA has repeatedly referred to kratom as a “new dietary ingredient” (NDI). In 1994, DSHEA established that substances marketed as dietary supplements before 1994 would not be subject to the NDI classification. If there is evidence that kratom was marketed as a dietary supplement before 1994, it would not be classified as an NDI. So far this evidence has not been found. Yet FDA has turned down multiple New Dietary Ingredient Notifications (NDIN) from suppliers, citing the applicants’ inability to demonstrate kratom as “reasonably expected to be safe”.
Unlike new drugs, dietary supplements are not reviewed and approved by FDA based on their safety and effectiveness. Unless an exception applies, dietary supplements that contain a new dietary ingredient (a dietary ingredient not marketed in the United States before Oct. 15, 1994) require a notification to FDA at least 75 days before marketing.
The notification must include the information that provides the manufacturer’s or distributor’s basis for concluding that the dietary supplement will reasonably be expected to be safe. When public health concerns arise about a dietary supplement after the product is on the market, FDA evaluates the product’s safety through research and adverse event monitoring.
https://www.fda.gov/consumers/consumer-updates/it-really-fda-approved
Given the FDA’s unwillingness to engage in a rational, evidence based discussion concerning kratom’s safety, obtaining NDI or Generally Recognized as Safe (GRAS) status is impossible without rational FDA leadership. Therefore, kratom is held in regulatory limbo by operatives in both government and the pharma/supplements industry who have a vested interest in roadblocking sensible kratom legislation, and who couldn’t give the first fuck about public health or your well-being.
If you want to take preaching from kratom prohibitionists in the pharma/supplements industry seriously, you have to forget about the fact that the dietary supplements industry has traditionally had massive ethical and safety issues in and of itself.
In 2011, NPA’s VP of Global Government and Scientific Affairs, Daniel Fabricant, passed through the revolving door to become Division Director for Dietary Supplement Programs at FDA. Three years later, through efforts by Fabricant at the urging of the pharma/supplements industry, FDA issued its first import alert against kratom on March 1, 2014.
That import alert gives the FDA the power to call the US Marshals away from important work, like chasing fugitives, to seize shipments of kratom that come into the US. The only thing keeping the FDA from seizing all kratom shipments is the lack of enforcement resources, which is why FDA sought and is still seeking to have the Drug Enforcement Administration (DEA) to place kratom on the list of Schedule I narcotics. That would give every cop in the country the power to seize all kratom everywhere.
Later in 2014, Fabricant left FDA to take a job as the NPA’s new Chief Executive Officer. Nothing to see here.
Timeline of NPA statements and actions against kratom
May 2011
Daniel Fabricant becomes FDA director of Dietary Supplements Program.
“It’s outrageous. It shouldn’t happen,” said Sidney Wolfe, the director of the health research group at the watchdog organization Public Citizen. Wolfe, a longtime critic of the FDA, said if the agency hired someone from a pharmaceutical trade group to oversee drug regulation “there would be a huge uproar and there should be here.”
https://archive.sltrib.com/article.php?id=51715033&itype=CMSID
June 11, 2012
First FDA import alert, 66-41, that includes kratom.
July 18. 2013
Other big pharma/supplements industry groups write a letter to Daniel Fabricant whining about kratom:
[Council for Responsible Nutrition] CRN, the United Natural Products Alliance (UNPA) and the Consumer Healthcare Products Association (CHPA) asked FDA to clarify its safety concerns over kratom and its position that the botanical is not a legitimate dietary ingredient.“Given the widespread availability of kratom, the dietary supplement industry is concerned about the potential dangers to consumers who may believe that they are consuming a safe, regulated product when they are not,” the trade groups wrote in a July 18, 2013 letter to Daniel Fabricant, Ph.D., FDA’s director of the division of dietary supplement programs.
https://www.naturalproductsinsider.com/regulatory/fda-states-mount-war-kratom
March 1, 2014
FDA issues Import Alert 54-15 giving it the power to use US Marshals to seize kratom products. This is the import alert updated yearly by FDA.
April 7, 2014
Fabricant leaves FDA to take a job as CEO of NPA.
October 13, 2014
FDA seizes $5 million worth of kratom from a Van Nuys, CA supplier.
June 2, 2016
NPA narcs on a kratom company for… selling kratom.
In June 2021, four years after his initial Freedom of Information Act request, journalist Anthony Roberts uncovered two emails dated June 2, 2016 sent from NPA to FDA, notifying the FDA that the company Happy Hippo Herbals was selling kratom. In an email to FDA’s Michael Cummins, NPA’s Corey Hilmas said, “Kratom has an import alert out for it and the agency has essentially banned it.” In an email to FDA’s Robert Durkin, NPA’s Joy Akins said, “Additionally, we wanted to bring to your attention to Red Supplements that is selling Kratom under the website happy hippo herbals.”
The emails made no mention of GMP compliance or testing of kratom products, only that this company sold kratom, which in the opinion of the NPA was illegal. There was no indication this company was selling contaminated or adulterated products. Happy Hippo Herbals has since become a member of the American Kratom Association’s GMP program.
August 31, 2016
In a press release (pdf) following the DEA’s announcement of intent to place kratom on Schedule 1 list, NPA called DEA’s action “toothless” without enforcement, and went on to make a statement that Scott Gottlieb would echo five years later:
“Kratom is not an herbal supplement: it is addictive, harmful, and worse, it may be contributing to America’s opiate epidemic. We are eager to work with the authorities and our members to help turn the tide against Kratom and ensure that it is seen as what the DEA says it is, a schedule I illegal narcotic that has no place in health or wellness.”
– Natural Products Association, 8/31/2016
November 18, 2016
November 29, 2016
In an press release (pdf) during DEA’s comment period on the intended scheduling of kratom, Fabricant ranted:
Excerpts from NPA’s Executive Director and CEO Dr. Dan Fabricant’s comments to DEA:
“Adding an untested and unregulated substance such as kratom to our food supply without the application of longstanding federal rules and guidelines would not only be illegal, it could likely be dangerous, leading to serious unintended consequences as our nation struggles with the crisis of opioid addiction.”
…
“As former Food and Drug Administration (FDA) officials and regulators of the dietary supplement industry, we can tell you that finished kratom products and raw kratom botanical ingredients have not met the strict standards products and new ingredients must adhere to in order to be marketed to the public and deemed safe for regular use in either our food or our drug supply.”“Self-medicating with kratom is dangerous without the necessary pre-market approval process set forth by our public health experts at FDA. Even if kratom were delivered as a food, kratom has never been filed as a new dietary ingredient to FDA’s Center for Food Safety and Applied Nutrition.”
– Natural Products Association, 11/29/16
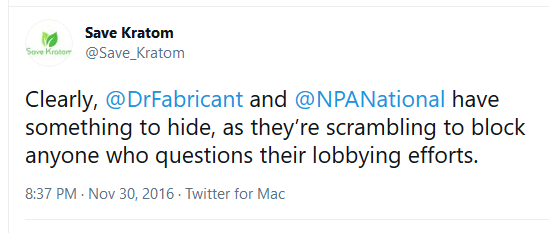
November 30, 2016
Fabricant and NPA block multiple kratom advocates on Twitter:

December 5, 2016
NPA press release (pdf) announcing Fabricant’s appearance on The Doctors daytime TV show:
“As the former top official overseeing the dietary supplement division at FDA, I can tell you that kratom has not met the legal and regulatory requirements to be sold on store shelves alongside products that have been proven to be safe such as vitamin C, fish oil supplements, or St. John’s Wart.”
– Natural Products Association, 12/5/16
Related “Fish oil or snake oil? Most capsules don’t contain what they promise” – TheConversation.com article from 2/18/2015
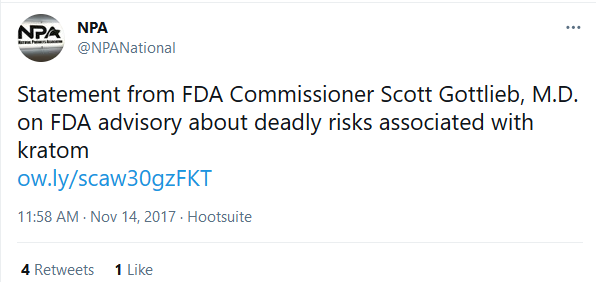
November 14, 2017
NPA posts Gottlieb statement about “deadly risks” of kratom.
December 1, 2017
Fabricant compares kratom to cocaine, from Natural Products Insider:
FDA has received three separate notifications in recent years to lawfully market kratom—the botanical from Southeast Asia—in dietary supplements, but the public health agency has objected to each submission based on determinations the notices failed to provide sufficient evidence of safety…
“Adverse event reports are rarely perfectly causal, but I think you start seeing signals stack up, which is what I think people have seen” with kratom, [Fabricant] said in a phone interview…
Added Fabricant: “There will never be an NDI just for kratom leaf in general. That would be the same as having an NDI for cocaine. That’s not going to happen.”
https://www.naturalproductsinsider.com/regulatory/fda-objections-kratom-dietary-supplements-highlight-safety-concerns
Or the same as having an NDI for coffee, if people had started drinking it after 1994.
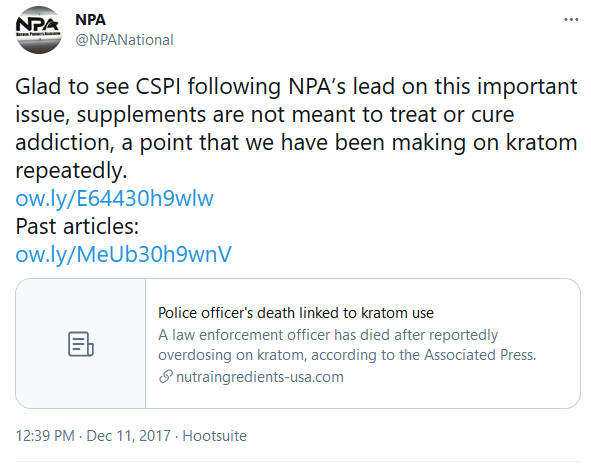
December 11, 2017
NPA tweets out a “kratom related death”
May 29, 2018
The kratom documentary A Leaf of Faith debuts on Netflix, written and directed by Chris Bell, who took on the dietary supplements industry in his previous documentary Bigger Stronger Faster. Fabricant appears as an anti-kratom voice in the documentary.
June 5, 2018
In an interview at Natural Products Expo West, Fabricant says he’s unaware of a proper NDIN or General Recognized as Safe (GRAS) status for kratom. He suggests removing the “addictive alkaloids or show they’re not addictive”. This despite the fact that FDA has never made a real case for proving the substance “is addictive”, or that he previously said kratom will never get an NDI.
May 16, 2019
Fabricant speaks at the FDA Dietary Supplement Public Meeting (pdf). Apparently now it’s not that kratom is unsafe, it’s just that there’s no NDI filed on it…Even though it’ll never have an NDI, like with cocaine.
There’s 900 NDIs filed and, Steve, I think, you know, I, I like the mathematical exercise. I don’t know that I agree with it, that 4,600 that should have filed, but okay, the bottom line is this, I don’t think that people are going to file unless there’s a strong enforcement component.
And it’s really simple. These are all enforcement components the Agency has. Some of them are actually NDIs. The claim on kratom, it’s not that it’s unsafe. It’s actually, it’s actually held because it doesn’t have an NDI, under 402(f)(1)(B)…
Well, it’s a technical adulteration. Failing to file is a technical adulteration. Not meeting GMPs is also a technical adulteration. So there’s an import alert for all products that don’t meet GMPs. It seems that there’s somewhat of a picking favorites of which part of the statute people choose to enforce or not enforce and I think that that should cause some concern here. And there’s Mitragynine speciose, and again, kratom, and again the kratom charge is failing to file an NDI. While there is information on the safety, it is that they didn’t file an NDI.
– FDA Public Meeting, 5/16/19
March 19, 2020
NPA urges FDA to enact import alerts, referencing the 2014 import alert against kratom.
June 3, 2020
NPA once again urges FDA to enact import alerts, referencing the 2014 import alert against kratom.
Fabricant added that the last time the FDA provided dietary supplement import alerts to prevent adulterated ingredients that have not completed the New Dietary Ingredient notification process entering the US was 2014 when he was director of the Division of Dietary Supplement Progrems (as it was then) for Kratom.
https://www.nutraingredients-usa.com/Article/2020/06/03/NPA-calls-for-FDA-to-issue-import-alerts-on-imported-adulterated-dietary-supplement-ingredients
July 20, 2020
NPA once again urges FDA to enact import alerts, referencing the 2014 import alert against kratom.
August 3, 2020
Fabricant talks about advocating for import alerts while he was in the FDA in an article in Natural Products Insider.
FDA issued an import alert on kratom, a botanical from Southeast Asia that FDA considers an NDI, when the leader of the Natural Products Association (NPA), Dan Fabricant, directed FDA’s then-Division of Dietary Supplement Programs.
Two import alerts currently apply to kratom, including one issued in 2019 (Import Alert 54-15) that remains in effect. Based on the agency’s “review of the publicly available information regarding kratom, there does not appear to be a history of use or other evidence of safety establishing that kratom will reasonably be expected to be safe as a dietary ingredient,” FDA stated in Import Alert 54-15.
In advocating for an import alert, Fabricant said his division approached other parts of FDA, including the Office of Compliance and Office of Chief Counsel, explaining justification for the requested action. Unlike a request for an injunction, an import alert does not require involvement from the U.S. Department of Justice (DOJ) and its attorneys, stated Fabricant, who led FDA’s supplement division from 2011 to 2014.
https://www.naturalproductsinsider.com/regulatory/fda-supplement-manufacturer-debate-ndi-import-alert-request
Nothing to see here.
###
*** In 2022, Fabricant was quoted as saying that he no longer supports kratom prohibition. To our knowledge, NPA has not made statements supporting kratom prohibition or kratom import alerts since 2020

Great write up and insight here properly focusing what went on.
Fabricant and Gottlieb are pharma/supp bros making a big $ play.
My husband is alive bc of kratom. He was choosing death over alcohol. He is a nonfunctional alcoholic.
He can now function like a normal person
Is fully sober with the use of kratom.
He can do his job, be an amazing dad and husband bc of this plant.
My brother in law would most likely still be alive if I knew of this plant.
He was given opiates for his ankle pain. He needed surgery and had pins. He got so hooked on these pills. The thought of surgery went out the window. Spent all of his money buying pills once he couldn’t get enough from the doctor. The doctor then just cuts you off and offers no help to help you stop the addiction they created. He went back amd forth btw pills and heroin. Heroin is cheaper than pills. Then died. He was only 30.
If I would have known about this plant and I would give anything to go back in time amd find this damn plant for him. It would have saved his life.
My husband after his brother passed is when his drinking went out of control.
He did rehab, AA, therapy, meds, outpatient, you name it, my hubby tried it.
I basically gave up all hope and was making sure if and when he died, that I knew what he wanted for burials. Made sure he was still paying life insurance etc.
I was also planning on getting a degree which I still am, so I could leave and be able to support my son and I. Now it can just be an extra income.
But I found kratom
Researched the hell out of it for months.
My husband never mentioned he had heard about it already. They bring it up in rehab and AA to stay away and never use, that it’s like pills or heroin.
🤦🏻♀️
Well what I read was another story. And this plant has been consumed for centuries. No deaths ever.
If there was a death with kratom in the system, the death is bc if other substances in the body and that is proven.
So I found where to buy this new plant online. Through a reputable company.
I have never looked back. My husband will now not die.
I use it rarely for energy, depression etc.
I took way too much to see what would happen. Again after research and watching leaf of life.
Well I got sick and puked. Slept it off. I have been 💯 worse with drinking a lot of alcohol while partying when younger.
Now alcohol before you get all sick is fun. If it wasn’t fun then no one would be getting sick from it. That’s until you are already addicted.
Now kratom, there isn’t anything fun about it. Kratom is something you could bring to a party and pass around. People would think you were nuts if you tried.
It’s a nice feeling… meaning some energy, not anything near cocaine, more like some really gooood coffee without jitters. And there are some strains of kratom that are just relaxing. Like one glass of wine but zero buzz.
Now if you take more than this, it just goes to being sick. And well you learn not to do that again and it’s just not worth it. With kratom, literally less is more. I can’t say that for really anything else.
So if you are against this plant, shame on you. Bc of the fear and lies spread about this plant bc if people spreading rumors being against it. Those people that could have tried kratom and were afraid to or if there is ever a ban….. those deaths are on you. And there are many. Sooooo many.
It should be common knowledge that kratom can help addiction, chronic fatigue, with knowledge of doctor of on medications, helps with anxiety and depression.
St. John worts you can not mix with antidepressants
They won’t tell you that in a store or even ask you if you buy them. You have to research that.
Kratom has not effected my medication but my doctor does know. I ended up talking to her about it.
This plant is amazing and has so much potential.
We need to find a way to use this against the FDA if they try again to schedule. I’m not certain how, but I think it can be done. I know the AKA already knows about it so they’d probably be the ones to spearhead it, but us advocates need to be involved too